Research
Stay up-to-date on the latest studies and bench-to-bedside therapies at Hopkins.
Thank You
Thank your interest. A staff member from the Clinical Research Office will contact you.
For any additional question, please contact our Clinical Research Office at 410-955-1167.
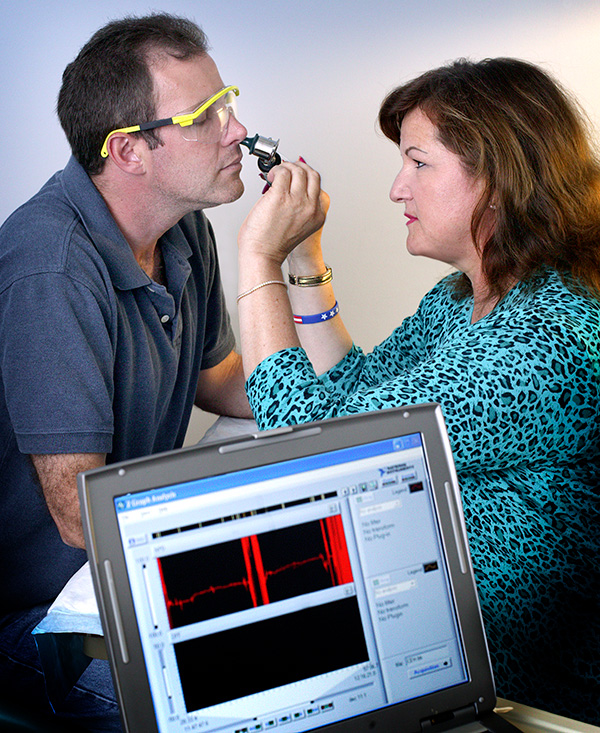
Nasal potential difference (NPD) measurement performed as part of a clinical trial.
Ongoing Trials
PI: Natalie West, MD, MHS
CONTACT: 410-955-1167
PROTOCOL: IRB00090863
The Standardized Treatment of Pulmonary Exacerbations II (STOP 2) is a randomized, controlled, open-label study being done to learn more about CF patients who have a pulmonary exacerbation treated with IV antibiotics. The main purpose of this research is to study the effectiveness and safety of differing durations of IV treatment, given in the hospital or at home for a pulmonary exacerbation in adults with CF.
People 18 years of age and older with CF who are experiencing a pulmonary exacerbation that their doctor wants to clinically treat with IV antibiotics may join. Participation in this study will last approximately 24 to 35 days and will include 3 study visits at Johns Hopkins. Procedures at the Study Visits may include: physical exam, vital signs, height and weight, blood draws, pulmonary function testing, and sputum collection. We are open for enrollment and expect to enroll up to 100 subjects into this study at Johns Hopkins.
You will be compensated for your time. If you expect to be treated with IV antibiotics for a pulmonary exacerbation at home or in
the hospital and are interested, please let us know! We can discuss the study further and answer any questions you may have.
PI: Peter J. Mogayzel, Jr. MD, PhD
CONTACT: Deanne Reyes at 410-955-1167
PROTOCOL: IRB00111304
Contact the Research TeamPI: Peter J. Mogayzel, Jr. MD, PhD
CONTACT: Deanne Reyes at 410-955-1167
PROTOCOL: IRB00147793
Contact the Research TeamPI: Natalie West, MD, MHS
CONTACT: Deanne Reyes at 410-955-1167
PROTOCOL: IRB00169474
Contact the Research TeamPI: Kiera A. Cohen, MD
CONTACT: Mary Rykiel at 410-955-1167
PROTOCOL: IRB00165735
Contact the Research Team